|
12/25/2023 0 Comments 3 dimensional shapes definition Molecules with More than One Central Atom Geometry of Molecules Chart Number of Electron Groups For example, a molecule with two bond pairs and two lone pairs would have this notation: AX 2E 2. The x represents the number of lone pairs present in the molecule. When lone pairs are present, the letter E x is added. "A" represents the central atom and n represents the number of bonds with the central atom. The VSEPR notation for these molecules are AX n. See the chart below for more information on how they are named depending on the number of lone pairs the molecule has.Īs stated above, molecular geometry and electron-group geometry are the same when there are no lone pairs. When the electron groups are all bond pairs, they are named exactly like the electron-group geometry. Molecular geometry, on the other hand, depends on not only on the number of electron groups, but also on the number of lone pairs. We separate this into two categories, the electron-group geometry and the molecular geometry.Įlectron-group geometry is determined by the number of electron groups. Although VSEPR theory predicts the distribution of the electrons, we have to take in consideration of the actual determinant of the molecular shape. Thus, the molecule's shape reflects its equilibrium state in which it has the lowest possible energy in the system. The electrons and the nuclei settle into positions that minimize repulsion and maximize attraction. The shape of a molecule is determined by the location of the nuclei and its electrons. Using the VSEPR theory, the electron bond pairs and lone pairs on the center atom will help us predict the shape of a molecule. An electron group can be an electron pair, a lone pair, a single unpaired electron, a double bond or a triple bond on the center atom. VSEPR focuses not only on electron pairs, but it also focus on electron groups as a whole. Thus, electron pairs will spread themselves as far from each other as possible to minimize repulsion. The valence-shell electron-pair repulsion (VSEPR) theory states that electron pairs repel each other whether or not they are in bond pairs or in lone pairs. Now that we have a background in the Lewis electron dot structure we can use it to locate the the valence electrons of the center atom. Valence-Shell Electron-Pair Repulsion Theory Therefore, the solid has 7 faces, 15 edges, and 10 vertices.\( \newcommand\) The edges intersect at 10 different points. The faces intersect at 15 different line segments. The solid has 2 pentagonal faces, on the top and bottom. The top view of a solid cylinder is in the form of a circle.Įxample 4: Find the number of faces, edges, and vertices of the solid figure given below: The front and the side views of the solid cylinder are both the same and are rectangular in shape. So, the given figure has 6 faces, 10 edges, and 6 vertices.ĭraw the front, the side and the top views of the solid shape given below.Įxample 3: Draw the top, the side and the front views of the solid cylinder. 
The edges intersect at 6 different points. The faces intersect at 10 different line segments. 
The figure has one pentagonal base and 5 triangular lateral faces. 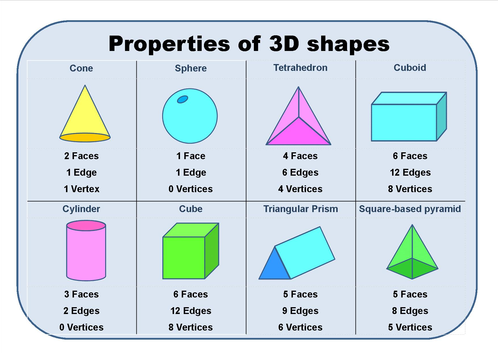 
Step 3: Mark any hidden edges as dashed lines.įinally, we obtain the three-dimensional triangular pyramid.įind the numbers of faces, edges, and vertices in the figure given below. Step 2: Connect all the three vertices of the triangle to the point. Step 1: Draw a triangle and mark a point above the triangle as shown in the figure. The following are the steps to draw a triangular pyramid on paper: Step 3: Draw the edges that are hidden or are on the backward side as dashed lines.įinally, the three-dimensional rectangular prism is obtained. Step 2: Connect the corresponding vertices using straight lines as shown. Step 1: Draw two identical rectangular faces as shown in the figure. The following are the steps to draw a rectangular prism on paper: We draw lines and curves on paper such that they can easily be visualised as three-dimensional solids. However, drawing three-dimensional figures on paper is quite different. We can easily draw two-dimensional figures on paper using a scale, pencils, a compass, and set squares. How can we draw three-dimensional figures? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
